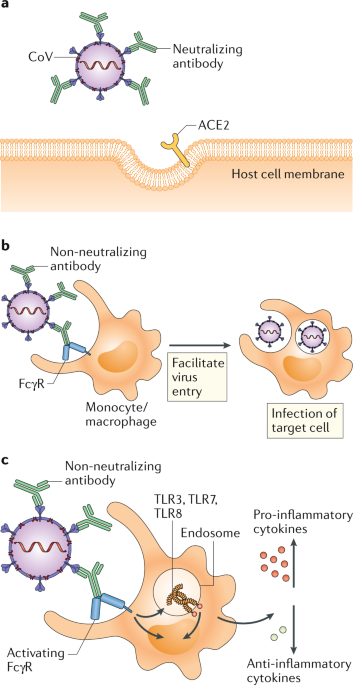
Multiple factors determine whether an antibody neutralizes a virus and protects the host or causes ADE and acute inflammation. These include the specificity, concentration, affinity and isotype of the antibody. Viral vector vaccines encoding SARS-CoV S protein and nucleocapsid (N) protein provoke anti-S and anti-N IgG in immunized mice, respectively, to a similar extent. However, upon re-challenge, N protein-immunized mice show significant upregulation of pro-inflammatory cytokine secretion, increased neutrophil and eosinophil lung infiltration, and more severe lung pathology8. Similarly, antibodies targeting different epitopes on the S protein may vary in their potential to induce neutralization or ADE. For example, antibodies reactive to the RBD domain or the HR2 domain of the S protein induce better protective antibody responses in non-human primates, whereas antibodies specific for other S protein epitopes can induce ADE10. In vitro data suggest that for cells expressing FcRs, ADE occurs when antibody is present at a low concentration but dampens at the high-concentration range. Meanwhile, increasing antibody concentrations promotes SARS-CoV neutralization by blocking viral entry into host cells6. For other viruses, high-affinity antibodies capable of blocking receptor binding tend to not induce ADE.
In the ‘multiple hit’ model of neutralization, the virus-blocking effect correlates with the number of antibodies coating the virion, which is collectively affected by antibody concentration and affinity11. Monoclonal antibodies with higher affinity for the envelope (E) protein of West Nile Virus (WNV) induced better protection in mice receiving a lethal dose of WNV11. For a given concentration of antibody and a specific targeting domain, the stoichiometry of antibody engagement on a virion is dependent on the strength of interaction between antibody and antigen. ADE is induced when the stoichiometry is below the threshold for neutralization. Therefore, higher affinity antibodies can reach that threshold at a lower concentration and mediate better protection11.
Antibody isotypes control their effector functions. IgM is considered more pro-inflammatory as it activates complement efficiently. IgG subclasses modulate immune responses via the engagement of different FcRs. Most FcγRs signal through ITAMs, but FcγRIIb contains an ITIM on its cytoplasmic tail that mediates an anti-inflammatory response. Ectopic expression of FcγRIIa and FcγRIIb, but not of FcγRI or FcγRIIIa, induced ADE of SARS-CoV infection6. Allelic polymorphisms in FcγRIIa are associated with SARS pathology, and individuals with an FcγRIIa isoform that binds to both IgG1 and IgG2 were found to develop more severe disease than individuals with FcγRIIa that only binds to IgG2 (ref.12).